Research News
More Evidence for Inflammation’s Role in Parkinson’s Disease- October 2019
New findings from a study being carried out by the Canadian LRRK2 in Inflammation Team (CLINT) are drawing connections between genetics, inflammation and the gut in Parkinson’s. Deeper understanding of the relationship between these pathways can help researchers better define, measure, and treat disease.
Scientists are constantly learning more about Parkinson’s disease:
- Mutations in the LRRK2 gene are a leading genetic cause of Parkinson’s and cause higher levels of the LRRK2 protein, which are also found in people with the disease without a genetic connection.
- Patients also show higher levels of inflammation in the brain. Inflammation is the body’s natural response to fight off infection, but too much or chronic inflammation can be harmful.
Finally, some have proposed that Parkinson’s starts outside the brain, accounting for changes in protein levels in the gut and olfactory bulb (responsible for smell) before motor symptoms begin.
A team of researchers from seven institutions across Canada- CLINT, published findings that connected these three processes last month in the journal Science Translational Medicine.
They found that disease models with a LRRK2 mutation had a greater inflammatory response to a bacteria and virus, but they also showed higher markers of inflammation in the brain, even though the virus and bacteria were not seen there. This work shows a role for the LRRK2 protein in modulating inflammation, but also that too much of a good thing may be harmful. The question of how the systemic inflammation leads to brain inflammation remains to be answered.
CLINT’s finding builds evidence for the Parkinson’s process beginning outside the brain. The gut and the nasal passage are often entry points for harmful things, so inflammatory response there may be an early trigger of the disease.
Other studies have shown an association between LRRK2 and inflammation, but this study builds on how they may be connected and points to new measurement and treatment strategies.
“If this theory about LRRK2 is correct, it could open the door for the monitoring of infections as a key risk element for prediction, early detection and prevention of Parkinson’s, and importantly, for new treatment approaches in general,” said senior author Dr. Michael Schlossmacher of The Ottawa Hospital.
As overactive LRRK2 has also been found in people with Parkinson’s who do not have a LRRK2 mutation, targeting this pathway may help a broader patient population. In fact, drugs against LRRK2 are already in clinical trials in patients with and without the mutation. This finding may have implications for that work as well.
“Our research suggests that these drugs may well succeed in safely reducing excessive inflammation”, said first author Dr. Bojan Shutinoski also of The Ottawa Hospital.
“However, we should be careful not to abolish LRRK2 function altogether, as this could make people more susceptible to infections, in particular when being treated potentially for years.”
Mindfulness Yoga Can Improve Anxiety and Depression in Parkinson’s
A Clinical Trial carried out by the University of Hong Kong in suggests that Mindfulness Yoga can improve anxiety and depression amongst people with Parkinson’s Disease.
Clinical practice guidelines support exercise for patients with Parkinson Disease (PD), but to our knowledge, no randomized clinical trials have tested whether yoga is superior to conventional physical exercises for stress and symptom management.
The objective of the 2017 trial was to compare the effects of a mindfulness yoga program vs stretching and resistance training exercise (SRTE) on psychological distress, physical health, spiritual well-being, and health-related quality of life (HRQOL) in patients with mild-to-moderate PD.
Mindfulness yoga was delivered in 90-minute groups and SRTE were delivered in 60-minute groups for 8 weeks.
Primary outcomes included anxiety and depressive symptoms assessed using the Hospital Anxiety and Depression Scale. Secondary outcomes included severity of motor symptoms (Movement Disorder Society Unified Parkinson’s Disease Rating Scale [MDS-UPDRS], Part III motor score), mobility, spiritual well-being in terms of perceived hardship and equanimity, and HRQOL. Assessments were done at baseline, 8 weeks (T1), and 20 weeks (T2).
Results revealed that the yoga group had significantly better improvement in outcomes than the SRTE group, particularly for anxiety and depression.
In conclusion, among patients with mild-to-moderate PD, the mindfulness yoga program was found to be as effective as SRTE in improving motor dysfunction and mobility, with the additional benefits of a reduction in anxiety and depressive symptoms and an increase in spiritual well-being and HRQOL.
For full information on the trial visit READ HERE

More Evidence that Parkinson’s Originates in the Gut -August 2019
Researchers at Johns Hopkins Medicine conducted studies in mice to investigate whether the toxic misfolded alpha-synuclein protein, which is a hallmark of Parkinson’s, could travel along the vagus nerve, which runs like an electrical cable between the stomach and small intestine into the base of the brain.
This study was prompted by previous research by German neuroanatomist, Heiko Braak, that showed people with Parkinson’s had a build-up of the misfolded alpha-synuclein in parts of the central nervous system that control the gut. Evidence from this latest study showed that alpha-synuclein began building where the vagus nerve connected to the gut and continued to spread through all parts of the brain.
It also demonstrated that blocking the transmission route could be key to preventing the physical and cognitive manifestations of Parkinson’s.
“These findings provide further proof of the gut’s role in Parkinson’s disease, and gives us a model to study the disease’s progression from the start… This is an exciting discovery for the field and presents a target for early intervention in the disease,” Says Professor Ted Dawson, M.D. Ph.D, Director of the Johns Hopkins Institute for Cell Engineering, Professor of Neurology at the Johns Hopkins University School of Medicine and a member of CPT’s Linked Clinical Trials committee
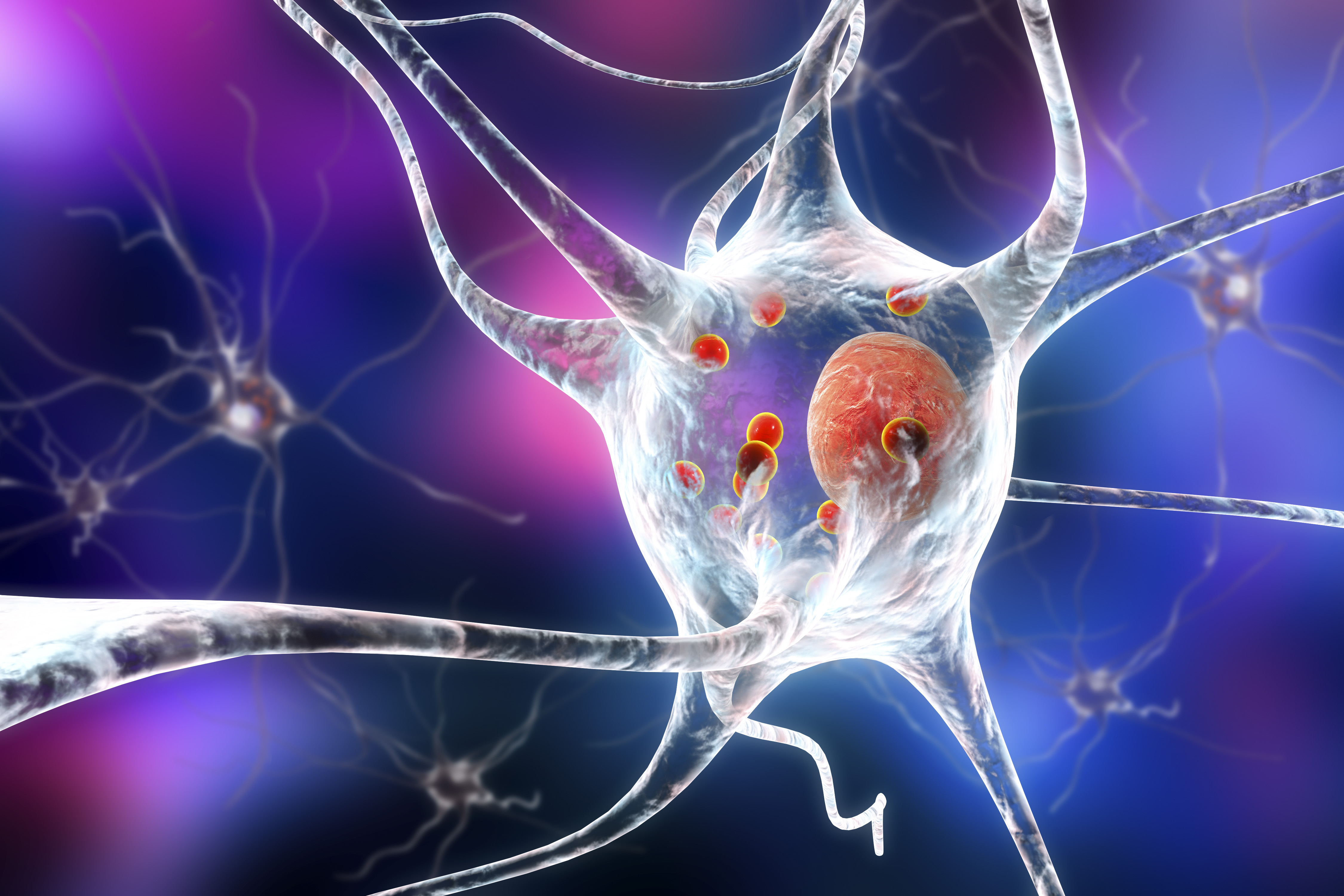
The Importance of Microglia in the Parkinson’s brain- August 2019
Microglia are a crucial population of helper cells whose primary function is to act as resident immune cells within the brain. They function like a ‘special force’, linked to the body’s immune system. They deal with injury/infection in the brain and communicate with the immune system about how best to cope with a developing condition. When microglia sense trouble in the brain, they will start to evaluate whether the affected cells need to be killed off or nurtured back to health.
Researchers at the Van Andel Institute in Grand Rapids (Michigan, USA) recently published a report demonstrating that microglia are significantly involved in the cell-to-cell transfer of the protein alpha-synuclein. This Parkinson’s-associated protein is believed to pass from one cell to another, and during this passage the alpha-synuclein begin to clump or aggregate within the cells. This process is how researchers believe the progressive nature of Parkinson’s is manifesting itself within the brain.
The Van Andel Institute researchers reported that the removal of microglia from the brain resulted in greater aggregation of alpha-synuclein, as did the hyperactivation of the microglia when a chemical used to induce inflammation was introduced. Their results suggest that under normal resting or ‘non-inflammatory’ conditions, microglia play an important role in modulating the transfer of alpha-synuclein in models of Parkinson’s.
Regulating microglia and neuroinflammation could represent a future avenue for limiting the progression of Parkinson’s – by reducing the spread of alpha-synuclein in the brain.
